With an average degree of polymerization of 8-100 units, paraformaldehyde is the smallest polyoxymethylene and a byproduct of formaldehyde polymerization. Due to breakdown, this material frequently has a mild formaldehyde odor. Due to its chemical and mechanical properties, paraformaldehyde takes various roles in the industry. As a disinfectant, hardness agent, and waterproofing agent, paraformaldehyde is employed. Additionally, it serves as an antiseptic in dentistry and is used to prepare adhesives and resins. Additionally, it is used as a thermoplastic.
Prior to delving into our insightful discussion about paraformaldehyde, it seems useful to bring up the name of a reputable manufacturer and supplier of this substance. JAM GROUP CO., as a prominent manufacturer, supplier and exporter of Chemical Auxiliaries and additives, is located in Iran. This country holds the fourth largest crude oil resource and the world’s second-largest natural gas resource. Iran also rates between the world’s top 10 oil producers and the world’s top 5 natural gas producers. JAM Group Co. was established by an experienced group of business and technical experts enjoying Iran’s rich resources and collaborating on providing quality products at the highest possible and consistent level. Paraformaldehyde is produced in our well-equipped factory and refinery in Alborz and Markazi provinces with cutting-edge technology that gives us exemplary power to fulfil our customer’s demands.
What is Paraformaldehyde?
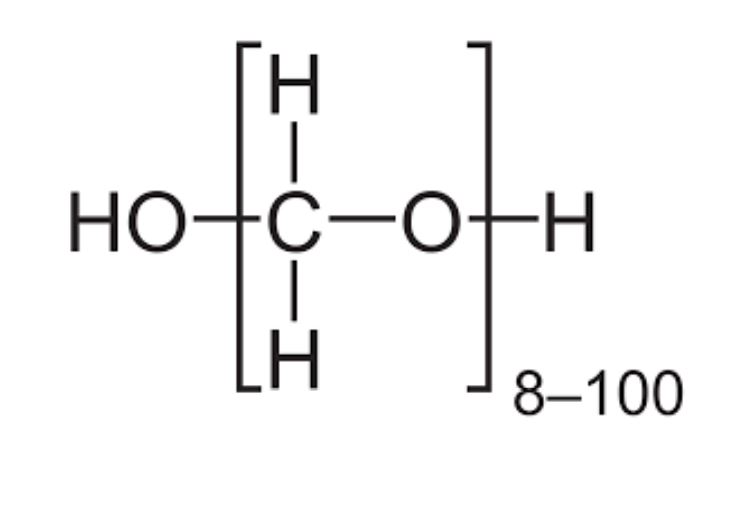
Paraformaldehyde (PFA) with the chemical formula OH (CH2O) NH (n=8-100), the unlimited solid form of formaldehyde, is a very useful reaction in classical organic reactions. Paraformaldehyde is the smallest polyoxymethylene, the polymerization product of formaldehyde with a typical degree of polymerization of 8 – 100 units. Paraformaldehyde usually has a lower odor than formaldehyde due to decomposition. Paraformaldehyde is a polyacetal, and has several applications in the chemical industry and chemical laboratories in organic chemistry.
The Properties of Paraformaldehyde
- Formula : OH (CH2O) NH (n=8-100)
- Density : 880KG/m3
- Melting point : 120°c
- Storage : store at<=20°c
- Iupac ID : Polyoxymethyle
- Appearance : white crystalline solid
- Solubility : Very low in water
- Flammable due to heating
A crucial note to consider is that paraformaldehyde reacts violently with strong oxidizers. It is also incompatible with inorganic acids and alkalis. Therefore it should be stored in a manner that would prevent the possible comingling with these compounds.
There are many other informal names, such as ‘paraform’, ‘formagene’, ‘para’. It is slowly formed as a white precipitate by condensation from the predominant species methanediol (formaldehyde hydrate) in solutions of formaldehyde (which may also be called ‘formalin’, ‘formal’, or ‘formalose’) on standing, in an equilibrium.
Formaldehyde is a gas. Its small molecules (HCHO, of which the -CHO is the aldehyde group) dissolve rapidly in water, with which they combine chemically to form methylene hydrate,
HO-CHj-OH. This is the form in which formaldehyde exists in aqueous solutions; its chemical reactivity is the same as that of formaldehyde. Methylene hydrate molecules react with one another, combining to form polymers. The liquid known as formalin contains 37-40% of formaldehyde and 60- 63% of water (by weight), with most of the formaldehyde existing as low polymers (n = 2 to 8). Higher polymers (n up to 100), which are insoluble, are used as a white powder, paraformaldehyde.
Different Grades of Paraformaldehyde
There are many chemicals in nature. Chemicals that are prepared by chemical synthesis and have a specific chemical composition have different grades. Including laboratory, food, industrial grade,etc .Of course, their chemical nature is very close to each other, but they differ in purity. In other words, the grade is the difference in the purity of chemicals for different uses.
Also, there are different grades because of the approvals that different organizations have given to the grades, The grade of chemicals is mainly divided as follows:
Reagent Grade
The purity percentage in this grade is 99%.
General Grade
According to the standards, the percentage of purity in this grade is different.
Laboratory Grade
The purity percentage is 97%.
Technical Grade
This grade has a low purity percentage and 90% impurity.
Among these, reagent grade is the purest and technical grade is the most miniature.
Other Grades of Paraformaldehyde
Paraformaldehyde is a product of concentrated methylene glycol, a substance that has not yet been isolated in its pure form. A white solid comes from perils or granules and contains between 78 and 98% formaldehyde. All forms include more than 95.5% paraformaldehyde. Granular forms with small, free, dust-free granules are popular with EM users. A fast-penetrating EM fixator used in combination with glutaraldehyde, acrolein and osmium tetroxide. There are more Paraformaldehyde grades in the market to fill the various needs for this substance; such as:
- Paraformaldehyde – methanol-free solution. A more efficient and rapid penetrant fixative is utilized in various Glutaraldehyde and Acrolein fixatives;
- Aqueous formaldehyde solution (aqueous paraformaldehyde solution) EM grade.
Paraformaldehyde in water is polymerized in a formaldehyde solution to produce stabilizing solutions of consistent quality. A water temperature of up to 60 ° C and an explanation of sodium hydroxide (added dropwise) are required to create a robust solution.
Paraformaldehyde Production Process
Paraformaldehyde is always manufactured from concentrated formaldehyde solutions. These are formed by low pressure distillation of the dilute formaldehyde solutions obtained in the conventional formaldehyde manufacturing plants. Paraformaldehyde is a mixture of polyoxymethylene glycols that contain about 8 to 100 formaldehyde units per molecule. The average degree of polymerization of paraformaldehyde varies with the conditions employed for the medication of the polymer.
The solution consists mainly of oligomers, but when n is large enough, the substance becomes insoluble enough to precipitate, while the concentration may continue. The resulting solid may have n ranging from ~ 8 to 100 or more. The reaction is directed to the left to release formaldehyde. It is also accelerated by low concentrations of formaldehyde, and in acidic or alkaline conditions. Solid paraformaldehyde smells like a monomer, and is therefore essentially a means of delivering formaldehyde slowly.
The paraformaldehyde is prepared from the concentrated solution, with 80-89 % of formaldehyde, in one of the following ways:
From a concentrated solution with 89-90% by weight of formaldehyde, paraformaldehyde is obtained by one of the following methods:
The First Method
Number one and the oldest way is making solid of thick formaldehyde solution in reaction vessel. This reaction is performed with or without a catalyst in a vessel equipped with a stirring blade to break up the mass formed. Or pouring the contents of the reaction container onto a cooled surface.
The Second Method
Alternatively, the concentrated solution is poured onto a heated roller, which evaporates part of the concentrated solution, resulting in a very pure formaldehyde product. ). This product is powdered, pelleted or flaky. These recently registered processes may also be continuous, but in all cases the final product must undergo a subsequent drying process to obtain a product that is hard enough not to form lumps during storage and transportation. In general, reaction vessels should be made of high molecular weight, highly acidic, and low-soluble products, or contain alkaline substances that cause depolymerization when dissolved in water.
The Third Method
Another method currently in use is the generalized process of a portable tower fed with a concentrated formaldehyde solution. The grains thrown from the tower are cooled by an air stream. This sequential process has many advantages over the previous ones. The product is obtained with a spherical physical shape, which is very useful for later uses. However, the newly produced material is still very soft and must mature later in the fluidized bed or on the conveyor belt.
The Fourth Method
In another modification of the process, the addition of reaction products between formaldehyde and an amine as the regulator of the molecular weight of the paraform is disclosed. This factor must be prepared in a separate process for 20 hours at 100°C. After its formation, it is added to a concentrated formaldehyde solution, which is crushed in a grinding tower. Here the product must be mature on a fluidized bed or on a conveyor for a certain period of time. Also, in low purity products (less than 89-87% by weight) the grains stick together and form lumps that do not move freely and become an unusable dough block.
About JAM Group Co.’s Paraformaldehyde
Various companies are in-demand for paraformaldehyde due to its role in diverse industries. It is vital to understand that using a high-quality grade of paraformaldehyde helps every producer improve outcomes with a noticeable utility. In this case, every company can rely on JAM Group Co, a prominent paraformaldehyde manufacturer and supplier. We at JAM Group count on the rich sources of oil and gas in Iran to organize a flawless supply chain with remarkable outcomes, such as high-quality paraformaldehyde. These products are produced in JAM Group’s factories in eastern Europe, Poland, and the Middle East. You can rely on the outcomes as certified products following international standards.
Moreover, every interested customer must know that JAM Group offers them various options related to packing and logistics. As usual, paraformaldehyde is packed considering universal rules to ensure transportation safety and easy filling. Still, please feel free to order your particular way of filling the material. In this case, do not hesitate to contact our German office experts; they may help you find the best way to arrange a shipment. you can also read more about this company, by clicking on About Us.
Conclusion
With a slightly unpleasant smell, paraformaldehyde is a white solid substance. It is a formaldehyde linear polymer with the formula HO(CH2-O)xH, where x is typically about 30. Due to its various properties, this substance is employed in multiple industries. As a disinfectant, hardness agent, and waterproofing agent, paraformaldehyde is operated. Additionally, it serves as an antiseptic in dentistry and is used to prepare adhesives and resins. Moreover, paraformaldehyde is used as a thermoplastic. This multipurpose product contains different grades that are used in distinct fields. Unique specifications produce each quality. Aqueous solutions of formaldehyde containing 37 or 55 per cent by weight are often the starting point for the production of paraformaldehyde. At lower temperatures, it is frequently concentrated by vacuum distillation, where initially, mostly water is distilled out.
| Property | Specification | Test Method |
| Chemical Type | Polyoxymethylene; polymerized formaldehyde (HCHO)ₙ, n ≈ 8-100 | – |
| Appearance | White to off‑white crystalline powder or prills; low odor | Visual inspection |
| Formaldehyde Content (as HCHO, w/w) | ≥ 90-97 % (after depolymerization) | Titrimetric oximation / iodometry (ISO 2227, adapted) |
| Free Formaldehyde (w/w) | ≤ 0.5-1.0 % | Spectrophotometric (acetylacetone / Hantzsch) |
| Water Content (w/w) | ≤ 0.50 % | ASTM D6304 (Karl Fischer) |
| Methanol (w/w) | ≤ 0.50 % | GC‑FID |
| Acidity (as formic acid, w/w) | ≤ 0.05 % | Alkalimetric titration / Ion chromatography |
| Ash (residue on ignition) | ≤ 0.05 % | Muffle furnace 600-800 °C |
| pH @25 °C (10% w/v solution) | 3.5-6.5 | ISO 10523 |
| True Density (solid) @25 °C | 0.86-0.90 g/cm³ (informational) | – |
| Melting Range | 115-125 °C | DSC (ASTM E794) |
| Bulk Density (loose powder) | 0.45-0.65 g/cm³ | ISO 3923‑1 |
| Particle Size (residue on 1.0 mm) | ≤ 5.0 w% | ISO 3310 (sieves) |
| Iron (Fe) | ≤ 5 mg/kg | ICP‑OES / ICP‑MS |
| Shelf Life | 12 months at ≤ 20 °C in sealed, moisture‑proof packaging | Retain re‑test (assay / pH / color) |
| Packing | 25 kg bags; 500-1,000 kg jumbo bags; lined fiber drums | – |
| Storage | Cool (≤ 20 °C), dry, well‑ventilated; keep sealed; segregate from strong oxidizers, acids and bases; avoid heat/ignition | – |
Paraformaldehyde is a compact form of formaldehyde, thus having similar properties, but with a wider range of applications. Paraformaldehyde is an antiseptic and disinfectant and acts as a source of formaldehyde. The resulting formaldehyde acts as an antibacterial, antifungal and antiviral, while it is less effective. It is also used to treat warts on the palms of the hands and feet and to flush out cavities from surgical hydatid cysts to eliminate scoliosis. Paraformaldehyde is very effective in disinfection and is entirely in the liquid formalin range. Because they do not need catalysts (permanganate), costs are reduced. They also take up less space in storage, and it is worth noting that the transport of paraformaldehyde is more cost-effective and effortless.
Paraformaldehyde Applications across the Industry
The application of paraformaldehyde in industries is varied because of its chemical and mechanical properties; below, you may discover a few of them:
- By duplicating it, it is used as a preservative.
- It is used in the process of cell culture.
- Paraformaldehyde is also used to bind proteins to DNA.
- As a sterilizer and disinfectant in the food, beverage, cosmetic and detergent industries due to its antibacterial properties,Paraformaldehyde is an antiseptic and disinfectants and acts as a source of formaldehyde. The resulting formaldehyde acts as an antibacterial, antifungal and antiviral, while it is less effective. It is also used to treat warts on the palms of the hands and feet and to flush out cavities from surgical hydatid cysts to eliminate scoliosis. Paraformaldehydes are very effective in disinfection and they are completely in the range of liquid formalin. Because they do not need catalysts (permanganate), they save money. They also take up less space in storage, and it is worth noting that the transport of paraformaldehyde is more cost-effective and easier.
- 97% As a rich source of formaldehyde in the production of a wide range of thermocyte resins
- Disinfectants, fungicides in the oil industry and its refining
- Starch fermenter in oil well drilling industry
- Paraformaldehyde is used in the production of 1, 3, 5-triazine in oil drilling.
- Prevent the growth of algae and microorganisms in the sugar industry
- Most paraformaldehyde is used by the chemical agro-industry for herbicides such as bismetriazole, butchloride, autochlorine, glyphosate, and machete.
- Used as a thermoplastic Paraformaldehyde is an alternative to aqueous formaldehyde for the production of resin, melamine, phenol
- Used in the production of chipboard, plywood and furniture in the wood industry
- Use in resins as casting powder
- Used as a resin in the paper and paper textiles industry
- Used as a dryer in the paint industry
- 96% powder paraformaldehyde/ 92%Prill paraformaldehyde. 92% paraformaldehyde is used as an ideal substitute for industrial formaldehyde and can be used in the manufacture of glyphosate, tricyclazole, trigger, stochloro as well as high-grade car paint, phenol formaldehyde resin, melamine resin and ion exchange resin, etc.
- As insulation for electronic components
- Use of paraformaldehyde in organic reactions with intermediate metals as catalyst
- In these methods, paraformaldehyde has been used as methylene blocks, hydroxylation ethylization reactions, CO source, gas substitute, hydrogen donor or receptor, formulation and methylation reactions.
- Aldehydes, such as formaldehyde, paraformaldehyde, and glutaraldehyde, are the fixatives of choice for standard morphologic analysis.
- The most common formaldehyde solution, known as formalin, is prepared with water and phosphate buffer to create 10% neutral buffered formalin (4% formaldehyde in phosphate-buffered saline). Formalin is the most generally used fixative for light microscopy and is a component in numerous other fixatives.
- Aldehyde fixatives cross-link proteins and are therefore known as cross-linking fixative
- Mixtures containing formaldehyde and glutaraldehyde
- The combination of formaldehyde with glutaraldehyde (as a stabilizer) in electron microscopy causes rapid penetration of small HCHO molecules and structural stabilization of the tissue. Rapid and thorough crosslinking is brought about by the more slowly penetrating glutadehyde oligomers.
- Reaction of formaldehyde with proteins
- The aldehyde group can combine with nitrogen and some other protein atoms, or with two atoms of this type (if they are very close together) to form a cross-linker -CH2- called the methylene bridge. Studies of the chemistry of tanning indicate that the most frequent type of cross-link formed by formaidehyde in collagen is between the nitrogen atom at the end of the side-chain of Iysine and the nitrogen atom of a peptide linkage. The production of leather with collagen tanning is comparable to the hardening of the tissue by the fixator.
- Formaldehyde, 10%, ultra-pure and without methanol, is suitable for both electron and optical microscopes. In large samples, it penetrates quickly but cross-linking slowly and easily. In combination with glutaraldehyde, it fixes delicate brain-like tissues in vascular perfusion
- Suitable for both electron and light microscopy.
- Paraformaldehyde is preferable to formalin when the tissue is prepared for electron microscopy. Because methanol added to formalin may coagulate cellular components and alter the delicate structure of tissue, subtle changes made by methanol are not visible on the surface of a light microscope. Paraformaldehyde is also preferred when briefly stabilizing tissue before use with labeled lectins because methanol appears to inhibit lectin binding.
Many applications, such as melamine-formaldehyde, urea-formaldehyde resins, or phenol-formaldehyde resins and adhesives for the production of particle board, do not require the pure paraformaldehyde obtained in portable towers to produce particle board. It is generally sufficient to be more than 80% by weight, which is sufficient for resin manufacturers to not have to pay 40% by weight to carry water in ordinary commercial formaldehyde solution, and also to distill excess water from the reaction.
○Why is formaldehyde often used in its solid Paraformaldehyde form?
Formaldehyde is a gas, making it difficult to handle and transport. Paraformaldehyde is its stable, solid polymer form, which acts as a convenient and safer way to store and use formaldehyde. When needed for a chemical reaction, the solid PFA can be gently heated or treated to release the formaldehyde monomer.
○How is Paraformaldehyde different from Formalin?
Formalin is the liquid solution, containing 37-40% formaldehyde gas dissolved in water where it exists as small polymers. Paraformaldehyde is the solid, higher-polymer version (up to 100 units long), which precipitates from the solution. PFA serves as a solid, stable source of formaldehyde, while formalin is the liquid version.
○How does Paraformaldehyde form?
Paraformaldehyde forms when formaldehyde molecules in an aqueous solution (like formalin) slowly react with one another. This condensation process links the individual methylene hydrate units together, forming long polymer chains. As these chains grow, they become insoluble in water and precipitate out as a white solid powder.
○What are the key safety considerations for storing Paraformaldehyde?
Paraformaldehyde is flammable when heated and reacts violently with strong oxidizers. It is also incompatible with inorganic acids and alkalis. Therefore, it must be stored in a cool location (at or below 20°C) and segregated from these other compounds to prevent hazardous reactions and ensure workplace safety.
○What form of Paraformaldehyde does JAM Group Co. supply?
JAM Group Co. supplies high-purity Paraformaldehyde as a white, free-flowing crystalline solid. Our product meets strict specifications for polymer length and purity, ensuring it serves as a reliable and consistent source of formaldehyde for various applications in the chemical industry and organic chemistry labs across the board.
○How does JAM Group Co. ensure the safe transport of Paraformaldehyde?
We prioritize safety in logistics. JAM Group Co. packages Paraformaldehyde in durable, sealed containers to prevent exposure to moisture and incompatible materials. Every shipment includes a Safety Data Sheet (SDS) detailing proper handling, storage requirements (≤20°C), and emergency procedures to ensure it arrives and is handled correctly.
HOW CAN JAM GROUP HELP YOU?
Send us a message and our friendly team will get back to you within one business day.
















