European alchemists first noticed soluble silicates of alkali metals (sodium or potassium) in the 1500s. Since then, these substances have played a vital role in various places. A common category of these chemicals is sodium silicate, the generic name for chemical compounds with unique formulas and multiple properties that lead to various utilities. There are many applications for the product, notably cement block manufacturing, fire suppression, textile and wood processing, as well as the manufacture of refractory ceramics, adhesives, and silica gels.
As a result, many industries must rely on superior grades of sodium silicate to produce stable products that meet market standards. JAM Group Co is one of the prominent manufacturers of sodium silicate in several qualities for industrial usage; every company worldwide can count on the JAM Group Co.’s sodium silicate as a premium product with highly robust properties. It is helpful to know that JAM Group Co.’s factories located in eastern Europe and the Middle East have enabled this sodium silicate manufacturer to present a product with international standards.
What is sodium silicate?
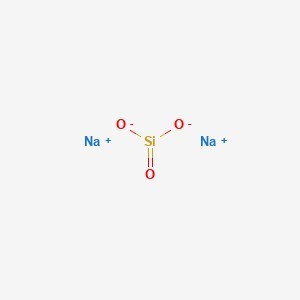
The term “sodium silicate” refers to a group of chemical substances containing the formula (Na₂O)ₓ·(SiO₂)ᵧ or Na₂ₓSiᵧO₂ᵧ₊ₓ, including sodium orthosilicate (Na4SiO4) and sodium pyrosilicate (Na6Si2O7). Anions are often polymeric. These substances often come in the form of white powders or colorless translucent solids that are soluble in varying degrees of water.
A combination of these substances, primarily the metasilicate, often known as water glass, water glass, or liquid glass, is also referred to both technically and colloquially as sodium silicate. The solution can be used in a wide range of applications, such as the creation of types of cement, passive fire protection, the processing of textiles and wood, and the fabrication of refractory ceramics, adhesives, and silica gel. The commercial product, which is available as a water solution or solid, is usually green or blue due to impurities containing iron.
Melting point 1089°C
Molecular Weight: 122.063 g/mol
BOILING POINT: 100°C
Chemical safety

Sodium silicate Ratio
Sodium silicate glass composition can be designated as: Na₂O·(SiO₂)ₓ where x is the ratio of the components and falls in the practical range from 0.4 to 4.0.
Different grades of the product
The majority of laboratory chemicals are labeled with a quality, which typically indicates the chemical’s purity. Although there are numerous standards used in grading, you are most likely to come across the ACS, Reagent Grade, and Laboratory Grades. Various chemical producers may employ different terms; however, as a general guideline, ACS and Reagent Grades are typically the purest, whereas Laboratory Grade is less pure. Chemicals that are ACS Grade are typically more expensive than chemicals that are lower grades because they are accompanied by a batch-specific Certificate of Analysis. As a middle-tier quality, laboratory-grade chemicals are frequently utilized in less thorough studies and educational labs.
As mentioned before, sodium silicate is a general name used to refer to a variety of chemical substances with different purities and chemical and mechanical properties. Each of these products can be introduced as different grades of sodium silicate. They may be differed based on variables below:
- Specific gravity;
- Na2O content;
- SiO2 content;
- Modulus (SiO2:Na2O);
- Total solids content.
In the industrial world, the weight ratio of SiO2 to Na2O distinguishes the different classes of sodium silicate. The ratio range is 1:2 to 3.75:1. Alkaline grades are those whose ratios are less than 2.85:1. They are referred to as neutral if their SiO2:Na2O ratio is higher.
Sodium silicate production process
Sodium silicate has been manufactured since the 19th century, and the basic principles of making “silicate of soda” have not changed since that time. It is commonly produced by roasting various quantities of soda ash (sodium carbonate, Na2CO3) and silica sand
(a ubiquitous source of SiO2) in a furnace at temperatures between about 1,000 and 1,400°C (approximately 1,800 and 2,500°F), a process that gives off carbon dioxide (CO2) and produces sodium silicate (Na2SiO3; usually represented by its two constituents, Na2O and SiO2):
Na2CO3 + SiO2→ Na2O∙SiO2 + CO2
This roasting produces fused glassy lumps called cullet, which can be cooled and sold in that form or ground up and sold as powders. Lump or ground water glass in turn can be fed into pressurized reactors for dissolving in hot water. The solution is cooled to a viscous liquid and sold in containers ranging in size from small jars to large drums or tanks
Sodium silicate liquid can also be prepared directly by dissolving silica sand under pressure in a heated aqueous solution of caustic soda (sodium hydroxide, NaOH):
2NaOH + SiO2 → Na2O∙SiO2 + H2O
In either production route, the higher the ratio of SiO2 to Na2O and the higher the concentration of both ingredients, the more viscous the solution. Viscosity is a product of the formation of silicate polymers, the silicon (Si) and oxygen (O) atoms being linked by covalent bonds into large negatively charged chain or ring structures that incorporate the positively charged sodium ions as well as water molecules. Highly viscous solutions can be spray-dried to form glasslike beads of hydrated sodium silicate. The beads can be packaged for sale to commercial users much like ground cullet, but they dissolve more readily than the anhydrous form of water glass.
About JAM Group Co.’s Sodium Silicate
In the previous section, various functions of sodium silicate were discussed. This multipurpose chemical compound is applied in diverse industries due to its properties. Every company must collaborate with a reliable sodium silicate manufacturer to obtain various grades of the product. JAM Group Co is a leading sodium silicate exporter that counts on its equipped factories and skilled chemists and provides you with multiple grades of this chemical compound. All outcomes of these factories are high-quality materials suitable for industrial applications in accordance with international standards.
With JAM Group Co., you have a variety of options when it comes to conducting business. This company offers you various choices for selecting the right product and logistics as a reputable sodium silicate supplier. We will be happy to deliver your desired grade of sodium silicate with your intended purity and additional properties. It is up to you to decide on your preferred packaging method with your chosen weight. Please do not hesitate to contact our experts in this case.
Conclusion
Sodium silicate is an inorganic sodium salt with silicate as the counterion. It has an ion of silicate in it. When solid, sodium silicate takes the form of a powdery or flaked material. This chemical compound has many different grades, each with a unique specific gravity, viscosity, and water concentration. It is helpful to know that water glass is created when silica sand and soda ash combine at a temperature of around 1,100 C. The water glass is crushed, dissolved in water, and digested under pressure with steam to create sodium silicate. The outcome can be engaged in numerous applications, including cement creation, passive fire protection, textile and wood processing, and refractory ceramics, adhesives, and silica gel production.
| Property | Specification | Test Method |
| Chemical Type | Aqueous sodium silicate (Na2O-SiO2 system); water glass; grade‑dependent modulus | – |
| Appearance | Colorless to pale‑straw, clear to slightly hazy viscous liquid; free of visible suspended solids | Visual inspection |
| Color (Gardner, neat) | ≤ 3-6 (grade‑dependent) | ASTM D1544 |
| Solids Content (wt%) | 35-55 | Gravimetric at 105 °C (ISO 787‑2) |
| Molar Ratio (SiO2:Na2O, modulus) | 2.0-3.5 (declare target) | Calculated from SiO2/Na2O assays |
| SiO2 (wt% of solution) | 20-40 (grade‑dependent) | ICP‑OES / wet chemistry |
| Na2O (wt% of solution) | 8-18 (grade‑dependent) | ICP‑OES / wet chemistry |
| Viscosity @25 °C | 50-1,000 mPa·s (grade‑dependent) | ISO 2555 (Brookfield) |
| pH @25 °C (10% w/w) | 11.0-12.7 | ISO 10523 |
| Density @25 °C | 1.35-1.60 g/cm³ (grade‑dependent) | ASTM D4052 |
| Insoluble Matter | ≤ 0.05 wt% | Filtration / gravimetric |
| Chloride (Cl⁻) | ≤ 100 mg/kg | Ion chromatography (IC) |
| Sulfate (SO4²⁻) | ≤ 200 mg/kg | Ion chromatography (IC) |
| Iron (Fe) | ≤ 30 mg/kg | ICP‑OES / ICP‑MS |
| Shelf Life | 12 months in unopened containers | Retain re‑test (assay/viscosity/pH) |
| Packing | 200 L drums; 1,000 L IBCs; bulk road/ISO tank | – |
| Storage | Cool, dry, covered; protect from CO₂ uptake and freezing; keep sealed and ventilated area | – |
Applying sodium siiste (SS) in practice allows for the more effective and efficient performance of a variety of good routine operations. All of this is made feasible by silicate colloid precipitation and polymerization. Silicate is a unique substance since it is water soluble, non-flammable, non-explosive, inorganic, non-toxic, economical, and environmentally benign. For example, adding SS to the drilling fluid allows problematic shales to be drilled more effectively than conventional oil-based muds (OBM), and lost circulation can be easily fixed using SS-based techniques. Below, you can discover the applications of sodium silicate more thoroughly.
Deflocculating silicates help break up inorganic or particulate soils into fine particles, making them easier to remove from surfaces and easier to suspend in solutions.
Adhesive
The most common application of sodium silicate solutions is in producing cardboard which is used as a paper cement
Drilling fluids
Sodium silicate is frequently used in drilling fluids for stabilizing borehole walls and avoiding the collapse of bore walls. It is particularly functional when drill holes pass through argillaceous formations containing swelling clay minerals such as smectite or montmorillonite.
Concrete and general masonry treatment
The use of sodium silicate solution in Concrete treatening could help to decrease porosity in most masonry products including concrete, stucco, and plasters. The consequence is in reducing water penetration, but has no known effect on reducing water vapor transmission and emission. A chemical reaction occurs with the excess Ca(OH)2 (portlandite) present in the concrete that permanently binds the silicates with the surface, making them much more durable and water repellent. This treatment commonly is used only when the initial cure has occurred (7 days or more depending on conditions). These coatings are known as silicate mineral paint.
Detergent auxiliaries
Sodium silicate is also used in detergent auxiliaries such as complex sodium disilicate and modified sodium disilicate. The detergent granules gain their ruggedness from a coating of silicates.
Water treatment
Sodium silicate is employed as an alum coagulant and an iron flocculants in wastewater treatment plants. Using Sodium silicate cause colloidal molecules blinds and leads to create aggregates that sink to the bottom of the water column. The microscopic negatively charged particles suspended in water interact with sodium silicate.as a result of increasing of ionic strength caused by the addition of sodium silicate their electrical double layer collapses and they subsequently aggregate. This process is called coagulation.
Refractory use
When blended with lightweight aggregates like vermiculite and perlite, water glass becomes a useful binder for solids. This mixture can be used to make hard, high-temperature insulation boards for refractories, passive fire protection, and other applications, such as moulded pipe insulation.
It can also produce high-temperature adhesives when mixed with finely divided mineral powders, like vermiculite dust. In the presence of this dust, the intumescence (the ability to swell with heat) disappears, and the water glass serves as a simple matrix. Because water glass is inexpensive and widely available, it is a favored material in many refractory applications.
Sand casting
It is utilized as a sand binder in the process of sand casting of iron or steel. It concede the rapid production of a strong mold, by passing CO2 through the mixture of sand and sodium silicate in the mold box, which hardens it almost immediately.
Dye auxiliary in hand dyeing with reactive dyes that needs a high PH to react with textile fiber, sodium silicate is used as a fixative Using. Firstly, the dye is applied to a cellulose-based fabric, including cotton or rayon, or onto silk, secondly it is let to dry, after which the sodium silicate is painted on to the dyed fabric, covered with plastic to retain moisture, and left to react for an hour at room temperature. in hand dyeing that needs a high PH to react with textile fiber, sodium silicate is used as a fixative.
Metal repair
Sodium silicate accompanying silicate, is used in muffler repair and fitting paste. dissolving in water, make both sodium silicate and magnesium to become as a form of a thick paste that is easy to apply. When the exhaust system of an internal combustion engine heats up to its operating temperature, the heat ejects all of the excess water from the paste.
The silicate compounds which are left over have glass-like properties, making a temporary, brittle repair.
Automotive repair
Sodium silicate another use is as an exhaust system joint and crack sealer for repairing mufflers, resonators, tailpipes, and other exhaust components, with and without fiberglass reinforcing tapes. In this application, , to make the sodium silicate “glued” joint opaque, the sodium silicate (60 – 70%) is generally mixed with kaolin (40-30%), an aluminum silicate mineral. The sodium silicate is the high-temperature adhesive; the kaolin serves simply as a compatible hightemperature coloring agent., though Some of these repair compounds also have glass fibers to enhance their gap-filling abilities and reduce brittleness.
Sodium silicate can be used to fill gaps within the head gasket. typically used on aluminum alloy cylinder heads, which are sensitive to thermally induced surface deflection. This can be caused by many things including head-bolt stretching, deficient coolant delivery, high cylinder head pressure, overheating, etc.
“Liquid glass” (sodium silicate) is added to the system through the radiator, and allowed to circulate. Sodium silicate is suspended in the coolant until it reaches the cylinder head. At 100 105°C (212-221°F), sodium silicate loses water molecules to form a glass seal with a remelt temperature above 810°C (1,490°F).
A sodium silicate which is a temporary form of repairment can last two years or longer. The repair occurs rapidly, and symptoms disappear instantly. This repair works only when the sodium silicate reaches its “conversion” temperature at 100 – 105°C. Contamination of engine oil is a serious possibility in situations in which a coolant-to-oil leak is present. Sodium silicate (glass particulate) contamination of lubricants is detrimental to their function.
Sodium silicate solution is used to inexpensively, quickly, and permanently disable automobile engines. Running an engine with about 2 liters of a sodium silicate solution instead of motor oil causes the solution to precipitate, catastrophically damaging the engine’s bearings and pistons within a few minutes. In the United States, this procedure was used to comply with requirements of the Car Allowance Rebate System (CARS) program.
Safe construction
A mixture of sodium silicate and sawdust has been used in between the double skin of certain safes. This not only makes them more fire resistant, but also makes cutting them open with an oxyacetylene torch extremely difficult due to the smoke emitted.
Crystal gardens
When crystals of a number of metallic salts are dropped into a solution of water glass, simple or branching stalagmites of colored metal silicates are formed. This phenomenon has been used by manufacturers of toys and chemistry sets to provide instructive enjoyment to many generations of children from the early 20th century until now.
Pottery
Sodium silicate is used as a deflocculate in casting slips helping reduce viscosity and the need for large amounts of water to liquidize the clay body. It is also used to create a crackle effect in pottery, usually wheel-thrown. A vase or bottle is thrown on the wheel, fairly narrow and with thick walls. Sodium silicate is brushed on a section of the piece. After 5 minutes, the wall of the piece is stretched outward with a rib or hand. The result is a wrinkled or cracked look.It is also the main agent in “magic water”, which is used when joining clay pieces, especially if the moisture level of the two differs.
Sealing of leaking water-containing structures
Sodium silicate with additives was injected into the ground to harden it and thereby to prevent further leakage of highly radioactive water from the nuclear power plant .
Food and medicine
While not actually a medical use, sodium silicate, and other silicates, are the primary components in “instant” wrinkle remover creams, which temporarily tighten the skin to minimize the appearance of wrinkles & under-eye bags. These creams, when applied as a thin film and allowed to dry for a few minutes, can present dramatic results. Unfortunately, it is not permanent, lasts few minutes or couple of hours. It works like water cement, once the muscle starts to move, it cracks and leaves white residues on the skin.
Food preservation
Waterglass has been used as an egg preservative with large success, primarily when refrigeration is not available. Fresh-laid eggs are immersed in a solution of sodium silicate. After being immersed in the solution they were removed and allowed to dry. A permanent air tight coating remains on the eggs. If they are then stored in appropriate environment, the majority of bacteria which would otherwise cause them to spoil are kept out and their moisture is kept in. According to the cited source, treated eggs can be kept fresh using this method for up to five months. When boiling eggs so preserved, the shell is no longer permeable to air, and the egg will tend to crack unless a hole in the shell is made (e.g. with a pin) in order to allow steam to escape.
Aquaculture
Sodium silicate gel is also used as a substrate for algal growth in aquaculture hatcheries.
○What is sodium silicate, and what are its common names?
Sodium silicate isn’t a single chemical but a group of related compounds with the general formula (Na₂O)ₓ·(SiO₂)ᵧ. In technical and common terms, a mixture of these substances, especially in a water solution, is referred to as water glass or liquid glass due to its syrupy consistency.
○What are the common industrial applications of sodium silicate?
Sodium silicate, or water glass, is incredibly versatile. It’s used as a high-temperature adhesive, a binder in cements and refractory ceramics, and for passive fire protection. In other industries, it’s used for processing textiles and lumber, and it serves as a key raw material in the production of silica gel.
○What does sodium silicate look like?
In its pure form, sodium silicate appears as a white powder or a colorless, glass-like solid. However, the common commercial product, often a liquid solution, is typically green or blue. This coloration is not inherent to the silicate itself but is caused by the presence of iron-containing impurities from manufacturing.
○What does it mean that sodium silicate anions are often polymeric?
This means that the silicate anions, which form the compound’s backbone, often link together to form long chains or complex networks rather than existing as simple, individual ions. This polymeric nature is what gives sodium silicate solutions their viscous, syrupy consistency, much like “liquid glass,” and contributes to its adhesive properties.
○What forms of sodium silicate does JAM Group Co. supply?
JAM Group Co. provides sodium silicate in the forms most convenient for industrial use. We offer it as a solid, glass-like lump or powder, as well as in its popular aqueous solution form, commonly known as “water glass.” Our products are available in various grades to suit different applications.
○How does JAM Group Co. ensure the quality of its sodium silicate?
We ensure quality by precisely controlling the ratio of sodium oxide to silica in our products, which dictates their physical properties. Each batch is tested for viscosity, density, and purity to guarantee it meets the specific requirements for our clients’ applications, whether for adhesives, cements, or other industrial uses.
HOW CAN JAM GROUP HELP YOU?
Send us a message and our friendly team will get back to you within one business day.
























