With the chemical formula Na2SO3, sodium sulfite (also known as sodium sulphite) is an inorganic substance. It is a white, water-soluble substance that holds its unique physical and chemical properties like other chemical compounds; it is helpful to know that this substance is available in the market under several grades and forms, varied in purity and property. Sodium sulfite is mainly employed in the pulp and paper sector. It has also been applied in different sectors as an oxygen scavenger agent in water treatment, a textile industry reducing agent, and a sulfonation reagent. So, it is a multifunction substance that can be engaged in various industries for different applications.
JAM Group Co. is a reliable sodium sulfite manufacturer and exporter you can trust for pure products. In JAM Group factories located in Eastern Europe and the Middle East, various advanced machines and smart tools are engaged to produce different grades of sodium sulfate with high purity and reliable properties. Customers can count on JAM Group as a reputable sodium sulfite supplier that will deliver their intended materials with dependable shipments to their desired locations worldwide. In this case, go ahead and check About Us.
What is sodium sulfite?
Sodium Sulfite is the inorganic compound and a white odorless crystal or powder with sulfurous salty taste it decompress when heated, moreover it sinks in water and dissolves slowly and also transported as a heptahydrate Na2SO3.7H2O. It is generally available in powder, crystalline, and tablet forms
Physical Properties of Sodium Sulfite
Formula: Na2SO3
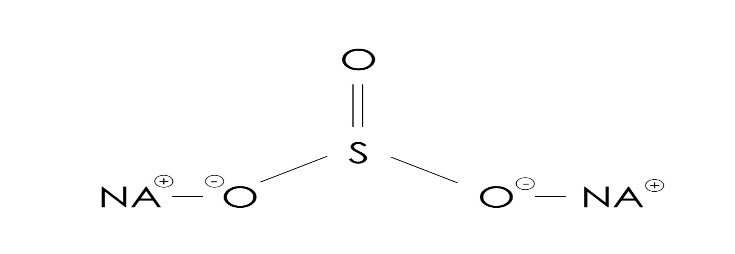
- Molar mass: 126.043 g/mol
- IUPAC ID: Sodium sulfite
- Density: 2.63 g/cm³
- The heptahydrate form density: 1.561 g/cm3.
When heated to 306.5K (33.4oC), the heptahydrate undergoes dehydration. The anhydrous form melts at a temperature of 500o Sodium sulfite does not have a specific boiling point since it tends to decompose at high temperatures. It is moderately soluble in water; its solubility corresponds to 27g/100mL. The crystal structure of anhydrous Na2SO3 is hexagonal whereas the heptahydrate crystals have a monoclinic structure Ph. 8.00-10.0
Chemical Safety:

Chemical Properties of Sodium Sulfite
When sodium sulfite contacts with strong or weak acids, Na2SO3 undergoes decomposition, liberating gaseous sulfur dioxide. Sodium sulfite reacts with aldehydes to yield a bisulfite adduct. although, sulfonic acids are produced from its reaction with ketones. Solutions of sodium sulfite are oxidized by atmospheric oxygen, yielding sodium sulfate. Sodium sulfite is not soluble in ammonia and chlorine.
Sodium sulfite production process
Sodium sulfite can be prepared by treating a solution of sodium hydroxide with sulfur dioxide. When conducted in warm water, Na2SO3 initially precipitates as a yellow solid. With more SO2, the solid dissolves to give the disulfite, which crystallizes by cooling.
Sodium Sulfite Preparation
1. In laboratories, sodium sulfite is generally prepared from the reaction between gaseous sulfur dioxide (SO2) and sodium hydroxide (NaOH). The chemical equation for this reaction is given by:
SO2 + 2NaOH → Na2SO3 + H2O
The depletion of the NaOH reactant can be detected via the addition of a few drops of concentrated H2SO4, concluding in the liberation of SO2 gas.
2. Industrially, Na2SO3 is produced from the reaction between sulfur dioxide and sodium carbonate solution. At first, sodium bisulfite (NaHSO3) is formed. This compound now reacts with sodium carbonate or sodium hydroxide to yield the sodium sulfite product. The reaction can be generalized to:
Na2CO3 + SO2 → Na2SO3 + CO2
Different grades of sodium Sulfite
Various items, including pharmaceuticals, medical devices, biologics, cell- and tissue-based products, and many other healthcare-related solutions, are manufactured and tested using chemicals and reagents, such as sodium sulfite. Laboratories, businesses, and researchers who utilize chemicals and reagents have confidence that their producers have accurately categorized each chemical’s grade and verified that it complies with all applicable legal requirements for its intended use. Everyone involved in the custodial supply chain must know the various chemical qualities and their purposes.
Like other chemical compounds, sodium sulfite is produced under various grades, each suitable for a particular purpose. Sodium sulfite manufacturers usually present this product in multiple grades, such as reagent, technical, and Food Chemical Codex. These products contain different amounts of purity. It is helpful to know that available sodium sulfite in the market contains 90-98% of Na2SO3.
About JAM Group Co.’s Sodium Sulfite
Why should we pick the JAM Group out of all the businesses operating in the sodium sulfite sector? There are many reasons to consider this company a trustworthy business partner. JAM Group’s sodium sulfide has distinctive properties. This product has neat sides, a translucent tint, and a well-defined shape. It is the ideal choice for applications in the mining and leather sectors.
It’s also important to note that working with JAM Group Co. gives you various logistic choices. Sodium sulfide is packaged in safe packings following international standards. Still, You can request specific filling ways. For instance, you can package your product in jumbo bags and alter the amount of each bag; these bags typically contain 1 ton of sodium sulfite, but customers can order them in 800-kilogram packs instead.
Conclusion
Sodium sulfite is an inorganic chemical compound with the formula Na2SO3. It is an odorless, solid, white powder that dissolves in water and tastes sulfurous and salty. Sodium sulfite producers usually present this substance under various grades, such as reagent, technical, and FCC; each holds multiple amounts of purity. These qualities are the outcomes of several production processes. Mainly, Sulfur dioxide and sodium carbonate (soda ash) are combined to create sodium sulfite, which is then purified and dried to produce crystals or powder. Numerous sectors employ sodium sulfite, including the pulp and paper sector, the photographic sector (to prevent developer solutions from oxidizing and to remove fixer from film and photo-paper), the textile sector, and the tanning of leather.
| Property | Specification | Test Method |
| Chemical Type | Inorganic sulfite; sodium sulfite (Na₂SO₃) crystalline powder/granular; anhydrous or heptahydrate | – |
| Appearance | White, free‑flowing crystals or powder; free of visible foreign matter | Visual inspection |
| Whiteness (index) | ≥ 85 (report method) | ASTM E313 / ISO 11475 |
| Assay (as Na₂SO₃, w/w) | ≥ 96.0-98.0 % (anhydrous basis; declare basis) | Iodometric titration / ICP‑OES |
| Moisture (w/w) | ≤ 0.50 % (anhydrous grade) | 105 °C oven (ISO 787‑2) |
| Sodium Sulfate (Na₂SO₄) | ≤ 1.0-3.0 wt% | Ion chromatography (IC) |
| Sodium Thiosulfate (Na₂S₂O₃) | ≤ 0.1-0.5 wt% | Iodometric / IC |
| Chloride (Cl⁻) | ≤ 0.05-0.20 wt% | Ion chromatography (IC) |
| Insoluble Matter | ≤ 0.05 wt% | Filtration / gravimetric |
| pH @25 °C (5-10% solution) | 8.5-10.5 | ISO 10523 |
| Density @25 °C | True density 2.6-2.7 g/cm³; bulk (loose) 0.80-1.20 g/cm³ | ISO 12154 / ASTM D1895 |
| Iron (Fe) | ≤ 10-50 mg/kg | ICP‑OES / ICP‑MS |
| Shelf Life | 24 months in dry, sealed packaging | Retain re‑test (assay / pH / insolubles) |
| Packing | 25 kg bags; 500-1,000 kg jumbo bags; palletized | – |
| Storage | Cool, dry, well‑ventilated; keep sealed; protect from moisture/CO₂; segregate from acids/oxidizers | – |
Sodium sulfite is mainly regarded as one of the Mineral Processing Chemicals. It is also used in photography, the bleaching of wool, and as preservatives in foods, beverages, and medications. it acts as useful antioxidant compounds and is also used in the manufacture of pulp for paper and wood products. Its preservative specifications include controlling microbial growth and the prevention of browning and spoilage.
Sodium sulfite application is as a sulfonation and sulfomethylation agent in the chemical manufacturing industry Sodium sulfite is primarily used as a food preservative (e.g., to prevent dried fruit from discoloring) and as an antioxidant. In the past, sodium sulfite was used to make pulp in the paper industry Sodium sulfite is also used water treatment and photography, Oil recovery, food preservatives, and coloring.
In Pulp and paper industry
Most sodium sulfite, 54%, is consumed in the pulp and paper industry for semi chemical and
acid sulfite pulping. The Kraft process ( sulfate process) is a process for conversion of wood into wood pulp, which consists of almost pure cellulose fibers, the main component of paper. The Kraft process entails treatment of wood chips with a hot mixture of
water, sodium hydroxide (NaOH), and sodium sulfide (Na2S), , that breaks the bonds that link lignin, hemicellulose, and cellulose. The technology entails several steps, both mechanical and chemical. It is the dominant method for producing paper.
In Food preservation
Sodium sulfite is often used to prevent the discoloration of dried fruits because it can act as a preservative. sodium sulfite, is used as preservatives in processed fruit products including dried fruits and juices, it is effective antioxidants that slow browning reactions and improve ascorbic acid retention in certain fruit products, It is used in the juice and juice beverage industry to inhibit malolactic bacteria, acetic acid bacteria, and spoilage yeasts and molds sodium sulfite demonstrates optimal antimicrobial effects below pH = 4 which recommend its use in high acid food systems such as fruit juices.
In Water Waste Treatment
One of the largest use of sodium sulfite is in water and wastewater treatment plants, where its application is to react with and neutralize excess chlorine used in the water and wastewater treatment processes.
In Photography
it is used in the developing process, and it acts as a preservative for the final picture produced.
In Swimming Pools
Sodium sulfite another application is in swimming pools to decrease the chlorine levels in the water.
As Oxygen Scavenger
The purpose of an oxygen scavenger is to limit the amount of oxygen available for deteriorative reactions that can lead to reduced functionality of many types of products, from pharmaceuticals to metals, to food This product is used in steam boiler water treatment to remove dissolved oxygen and to remove excess chlorine from wastewater. In boiler systems, this compound works as an oxygen scavenger to protect the system from pitting corrosion. so the water fed to boilers is often treated with sodium sulfite.
In Textile Industry
The textile industry makes extensive use of Na2SO3 for bleaching, dechlorinating and desulfurizing purposes.
In Purifying Trinitrotoluene
Sodium Sulfite is also used in the process of purifying trinitrotoluene (TNT) in order to make it fit for military use.
In Pharmacology
It is a useful agent in many drugs, which helps maintain their potency and stability.
○What is sodium sulfite and what are its common forms?
Sodium sulfite is an inorganic salt with the formula Na₂SO₃. It’s a white, odorless powder with a salty, sulfurous taste. It’s commonly available in an anhydrous (dry) form but is often transported and used as a heptahydrate (Na₂SO₃·7H₂O), which is a crystalline solid containing seven water molecules.
○What are the key physical properties of sodium sulfite?
The anhydrous form of sodium sulfite has a density of 2.63 g/cm³ and a hexagonal crystal structure. The heptahydrate form is less dense and has a monoclinic structure. Sodium sulfite is moderately soluble in water but decomposes at high temperatures instead of boiling, making it useful in specific thermal processes.
○What happens when sodium sulfite is exposed to acids or air?
When sodium sulfite comes into contact with acids, even weak ones, it decomposes and liberates sulfur dioxide gas. In solution, it’s also slowly oxidized by atmospheric oxygen, which converts it into sodium sulfate. This reactivity is a key consideration for its storage and use in various chemical applications.
○How does sodium sulfite react with aldehydes and ketones?
Sodium sulfite exhibits distinct reactions with different organic compounds. It reacts with aldehydes to form a bisulfite adduct, a common reaction in organic synthesis and purification. With ketones, however, it follows a different pathway, reacting to produce sulfonic acids, demonstrating its versatility as a chemical reagent.
○In what forms does JAM Group Co. supply sodium sulfite?
JAM Group Co. provides sodium sulfite in several forms to meet diverse industrial needs, including powder, crystalline, and tablet formats. We supply both the anhydrous and the stable heptahydrate (Na₂SO₃·7H₂O) versions, ensuring our clients receive the appropriate grade and form for their specific application.
○How does JAM Group Co. recommend storing sodium sulfite?
To prevent oxidation to sodium sulfate and decomposition from ambient moisture or acids, JAM Group Co. recommends storing sodium sulfite in tightly sealed containers in a cool, dry place. Proper storage is crucial to maintain the product’s purity and prevent the unintended release of sulfur dioxide gas.
HOW CAN JAM GROUP HELP YOU?
Send us a message and our friendly team will get back to you within one business day.




























